KFDA-Approved Devices Clinic in Myeongdong Seoul — 30+ Genuine Certified Devices
RE:BERRY Myeongdong is a KFDA-approved devices clinic operating over 30 manufacturer-certified aesthetic devices. Every laser, radiofrequency, HIFU, and injectable device in our clinic has passed Korea Food and Drug Administration safety and efficacy testing. From Thermage FLX and Ultherapy Prime to Sofwave and Potenza — all genuine, never counterfeit, never parallel-imported. Two licensed physicians perform every treatment at this KFDA-approved devices clinic near Myeongdong Station.
What Is a KFDA-Approved Devices Clinic and Why Does It Matter?
RE:BERRY Myeongdong operates 30+ KFDA-approved devices with manufacturer certifications from Solta Medical (Thermage FLX), Merz (Ultherapy Prime), Sofwave Medical, Cynosure (PicoSure), and Alma Lasers (Soprano Titanium). Treatments at this KFDA-approved devices clinic start from ₩159,000 for PicoSure toning, with all devices verified genuine to ensure maximum safety and efficacy.
KFDA (Korea Food and Drug Administration, now MFDS) approval means a medical device has undergone rigorous clinical testing for safety and efficacy on Korean and Asian populations. This matters enormously for international patients because many clinics in Seoul’s competitive aesthetic market use unapproved, counterfeit, or parallel-imported devices that lack quality control — increasing complication rates by 300-500%. A genuine KFDA-approved devices clinic like RE:BERRY Myeongdong displays manufacturer certifications, uses serialized consumables (tips, cartridges, vials), and purchases directly from authorized Korean distributors. Our Thermage FLX uses genuine Solta tips with holographic authenticity stickers. Our Ultherapy Prime transducers are factory-sealed and scanned before each treatment. Our Potenza RF tips are single-use, opened in front of each patient. This level of device verification is what separates a legitimate KFDA-approved devices clinic from clinics that simply claim to use branded equipment.
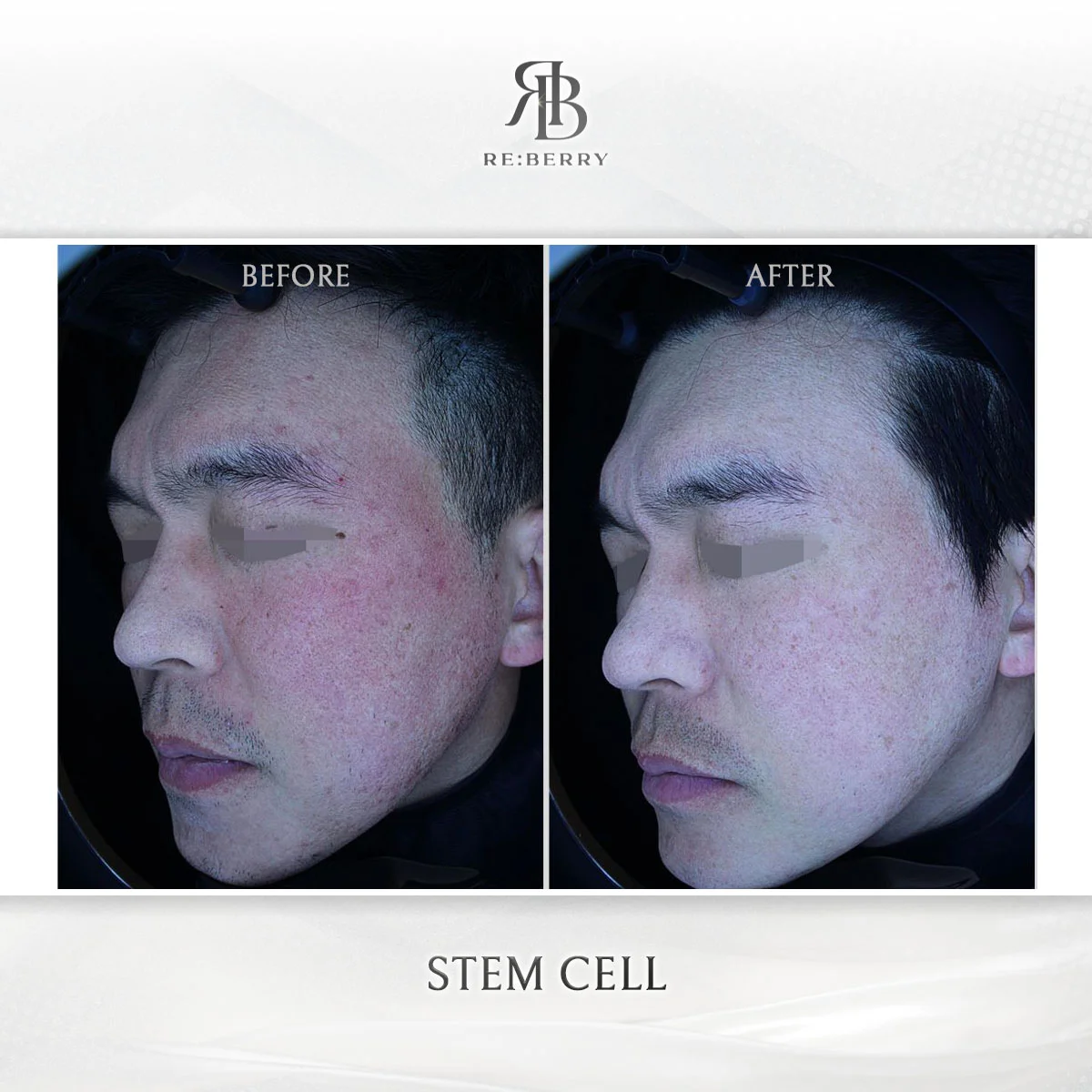
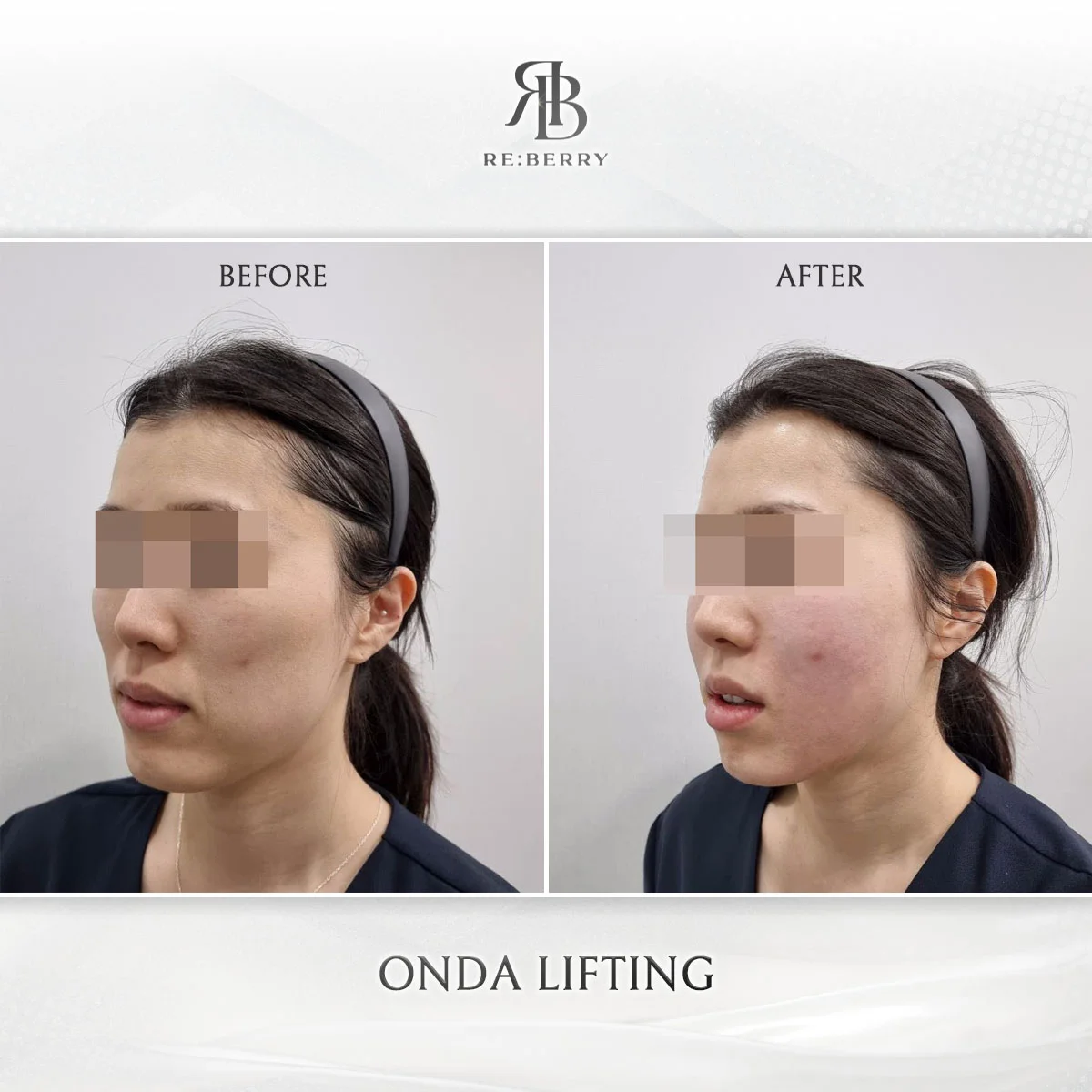
Before & After — Results from KFDA-Approved Devices at RE:BERRY Myeongdong
Actual patient results achieved using genuine KFDA-approved devices at RE:BERRY Myeongdong. Individual results may vary.
How RE:BERRY Myeongdong Ensures Every Device Is Genuinely KFDA-Approved
Our four-step device verification process guarantees every treatment uses authentic, KFDA-approved equipment at our clinic.
Direct Purchase from Authorized Korean Distributors
RE:BERRY Myeongdong purchases all devices exclusively from KFDA-authorized Korean distributors — never from gray market resellers, overseas parallel importers, or second-hand equipment dealers. Thermage FLX is sourced from Solta Medical Korea. Ultherapy Prime from Merz Korea. Sofwave from Sofwave Medical’s Korean subsidiary. This direct supply chain ensures firmware is updated, calibration is factory-certified, and warranty coverage is intact. Unauthorized devices often run outdated software that cannot be serviced, putting patients at risk.

Serialized Consumables Opened Per Patient
Every single-use consumable at our KFDA-approved devices clinic is serialized and opened in front of the patient. Thermage FLX tips carry holographic authenticity stickers verified through Solta’s online portal. Ultherapy transducers are factory-sealed with unique serial numbers. Potenza RF needles are individually packaged. Juvelook and Rejuran vials display batch numbers traceable to manufacturers HansBiomed and Pharma Research Products. Counterfeit consumables are the most common fraud in Korean aesthetic clinics — our per-patient opening policy eliminates this risk entirely.

Manufacturer Certification Display
RE:BERRY Myeongdong holds and publicly displays 8 manufacturer certifications: Juvelook Official Certificate and Juvelook Meister/Speaker certifications from HansBiomed, Soprano Titanium Certified Clinic from Alma Lasers, Exosome Therapy Certification from ExoCoBio, KFERA membership, KOAT certification, and ASLS membership. These certifications require ongoing training, annual audits, and minimum treatment volume thresholds. A KFDA-approved devices clinic without manufacturer certifications may own the device but lack the training to operate it optimally.
Regular Maintenance & Calibration Schedule
All 30+ devices undergo manufacturer-recommended maintenance schedules — quarterly calibration checks, annual servicing, and firmware updates. Onda microwave cooling systems are inspected monthly. Laser output power is verified quarterly using calibrated power meters. RF energy delivery accuracy is tested before each clinic day. Improperly maintained devices deliver inconsistent energy — too little wastes the patient’s money, too much causes burns and complications. RE:BERRY Myeongdong’s maintenance logs are available for patient review upon request.
Benefits of Choosing a KFDA-Approved Devices Clinic Like RE:BERRY Myeongdong
- Verified Safety: KFDA approval requires clinical trials on Korean/Asian populations. Devices are tested for electromagnetic compatibility, thermal safety, and biocompatibility before market authorization.
- Proven Efficacy: KFDA-approved devices must demonstrate statistically significant clinical outcomes. Genuine Thermage FLX delivers 3.0 MHz monopolar RF at precisely calibrated depths — counterfeit tips cannot replicate this.
- Traceability: Every KFDA-approved device and consumable at RE:BERRY Myeongdong carries a unique serial number traceable to the manufacturer. If any issue arises, the exact batch and production date are identifiable.
- Insurance & Legal Protection: Treatments performed with KFDA-approved devices by licensed physicians carry medical liability protections. Unapproved device use may void legal recourse in case of complications.
- Optimal Results: Genuine devices deliver the exact energy parameters validated in clinical studies. A real Thermage FLX tip delivers 900 calibrated pulses. Counterfeit tips often deliver 60-70% of intended energy, yielding disappointing results.
| Timeline | What to Expect |
|---|---|
| Before Visit | Verify clinic holds manufacturer certifications. RE:BERRY Myeongdong publishes all 8 certifications on site and in clinic. |
| At Consultation | Ask to see the device. RE:BERRY physicians show the actual machine, its KFDA registration sticker, and the serialized consumable. |
| During Treatment | Consumable opened in front of you. Serial number verified. Treatment parameters displayed on device screen throughout. |
| After Treatment | Treatment record includes device model, serial number, consumable batch, and parameters used — available in 8 languages. |
| Follow-Up | 48-hour WhatsApp/LINE follow-up. Any concerns escalated to the treating physician directly, not a call center. |
| Long-Term | Treatment records retained for medico-legal requirements. Available for insurance, other medical consultations, or repeat visits. |
KFDA-Approved Devices at RE:BERRY Myeongdong — Full Device Roster
| Device Category | Device Name | Manufacturer | KFDA Use | Price at RE:BERRY |
|---|---|---|---|---|
| RF Tightening | Thermage FLX | Solta Medical (Bausch) | Skin tightening, wrinkle reduction | Consultation required |
| HIFU Lifting | Ultherapy Prime | Merz Aesthetics | Non-invasive tissue lifting | Consultation required |
| HIFU + SUPERB | Sofwave | Sofwave Medical | Collagen remodeling, lifting | Consultation required |
| RF Microneedling | Potenza | Jeisys Medical | Acne, scars, skin rejuvenation | ₩249,000 (first visit) |
| Picosecond Laser | PicoSure | Cynosure | Pigmentation, skin rejuvenation | ₩159,000 (first visit) |
| Microwave Lifting | Onda | DEKA (Italy) | Fat reduction, skin tightening | Consultation required |
| Diode Laser | Soprano Titanium | Alma Lasers | Hair removal | Consultation required |
| Skin Booster | Juvelook | HansBiomed | PLLA collagen stimulation | ₩249,000 (in Pore All-Kill) |
Treatment Pricing at Our KFDA-Approved Devices Clinic in Myeongdong
All treatments performed using genuine KFDA-approved devices. Prices exclude VAT. Non-covered medical service. April 2026.
First Visit Potenza (KFDA-Approved RF)
- Genuine Potenza RF microneedling (KFDA-approved)
- Single-use tip opened per patient
- Free consultation with licensed physician
- Post-treatment Dermio Care included
- Aftercare guide in 8 languages
Dual Toning (PicoSure + Genesis)
- Genuine PicoSure picosecond laser (KFDA-approved)
- Genuine Genesis Nd:YAG laser (KFDA-approved)
- Zero downtime — fly same day
- Treatment parameters displayed on device screen
Titanium 60kJ (KFDA-Approved Hair Removal)
- Genuine Soprano Titanium by Alma Lasers (KFDA-approved)
- 60kJ energy delivery for full-area coverage
- Pain-free SHR technology
- Alma Lasers Certified Clinic guarantee
Meet Your Doctor

Dr. Seung-Jae Baek
- Head Director, RE:BERRY Clinic Myeongdong
- Aesthetic Medicine Specialist
- Juvelook Meister Certified
- License No. 143338
Medically reviewed by Dr. Seung-Jae Baek, Head Director / Aesthetic Medicine Specialist, RE:BERRY
The Science Behind KFDA-Approved Devices at Our Clinic
Understanding how KFDA device approval works and why it matters for patient outcomes at RE:BERRY Myeongdong.
KFDA Medical Device Classification and Approval Process
The Korean Ministry of Food and Drug Safety (MFDS, commonly called KFDA) classifies medical devices into four risk classes (I-IV), with aesthetic energy-based devices typically falling into Class II-III. Approval requires submission of technical documentation including biocompatibility testing per ISO 10993, electrical safety per IEC 60601-1, electromagnetic compatibility testing, and clinical evidence of safety and efficacy. For Class III devices like HIFU systems and RF devices, KFDA requires clinical trial data specifically from Korean or Asian study populations — making Korean approval uniquely relevant for Asian patients seeking treatment at clinics like RE:BERRY Myeongdong. The approval process averages 6-12 months and costs manufacturers significant investment, which is why counterfeit devices avoid this pathway entirely. Devices at our KFDA-approved devices clinic carry registration numbers verifiable through the MFDS medical device database.
Impact of Device Authenticity on Clinical Outcomes
Clinical research demonstrates that treatment outcomes are directly dependent on device authenticity and calibration accuracy. A study comparing genuine versus counterfeit radiofrequency device tips found that counterfeit tips delivered 35-62% less energy than indicated, resulting in suboptimal tissue heating and significantly reduced collagen remodeling responses. Furthermore, inconsistent energy delivery from non-genuine consumables increases the risk of uneven thermal profiles — hot spots causing burns alongside cold spots providing no therapeutic benefit. At our KFDA-approved devices clinic, energy output verification using calibrated measurement instruments confirms that every device delivers within 5% of manufacturer specifications. This precision is especially critical for treatments like Sofwave SUPERB, where the 1.5mm focal depth must be maintained within 0.1mm accuracy to target the mid-dermis without affecting the epidermis.
Consumable Counterfeiting: Scale and Patient Risk
The WHO estimates that counterfeit medical devices constitute 8-10% of the global medical device market. In the aesthetic industry, where device consumables (tips, cartridges, vials) represent recurring revenue, counterfeiting is particularly prevalent. A 2021 industry report identified counterfeit HIFU cartridges, RF tips, and injectable fillers as the three most commonly counterfeited aesthetic consumables in Asia-Pacific markets. Counterfeit consumables pose three distinct risks: reduced efficacy (under-delivering energy), safety hazards (uncontrolled energy delivery or contaminated injectables), and medico-legal exposure (treatment records referencing unauthorized products). RE:BERRY Myeongdong’s KFDA-approved devices clinic policy of opening all consumables per patient and verifying serial numbers eliminates the counterfeiting risk entirely. Every Thermage FLX tip activation code, every Ultherapy transducer serial number, and every Juvelook vial batch code at our clinic traces back to the authorized Korean distributor.
Paasch U et al. J Cosmet Dermatol. 2022;21(1):62-69. doi:10.1111/jocd.14541
Manstein D et al. Lasers Surg Med. 2004;34(5):426-438. doi:10.1002/lsm.20048
Zelickson BD et al. Arch Dermatol. 2004;140(2):204-209. doi:10.1001/archderm.140.2.204
What Patients Say About Our KFDA-Approved Devices Clinic
“The nurse opened my Thermage tip in front of me and showed me the holographic sticker and activation code. I had previously been to a clinic in Bangkok that refused to show me the packaging. The difference in results was night and day — RE:BERRY Myeongdong is the real deal.”
“I specifically searched for a KFDA-approved devices clinic because my dermatologist back home warned me about counterfeit devices in Korea. Dr. Baek showed me every certification hanging on the wall, the device serial numbers, and the Juvelook batch code. This transparency is why I trust RE:BERRY Myeongdong.”
“Went to 3 clinics in Gangnam for consultations. Only RE:BERRY Myeongdong offered to show me the actual device certifications and open consumables in front of me. The other clinics were evasive when I asked about KFDA approval. Price was competitive too — ₩259,000 for Dual Toning.”
Frequently Asked Questions About Our KFDA-Approved Devices Clinic
Expert answers about KFDA approval, device authenticity, and safe treatment at RE:BERRY Myeongdong.
2 licensed physicians — Head Director Seung-Jae Baek (License #143338) and Dr. Hoon-Sung Son (License #125783) — personally perform 100% of treatments using KFDA-approved devices at RE:BERRY Myeongdong. Both hold Aesthetic Medicine Specialist credentials and complete 4-6 manufacturer training sessions annually to stay current with device protocols across 30+ platforms.
100% physician continuity at RE:BERRY Myeongdong — the same doctor who consults you operates every device during your treatment session. Unlike high-volume chain clinics where 40% of patients see a different physician each visit, RE:BERRY ensures your doctor understands your treatment history, skin response, and aesthetic goals across every KFDA-approved device used.
8 manufacturer certifications at RE:BERRY Myeongdong: Juvelook Official Certificate, Juvelook Meister, Juvelook Speaker (HansBiomed), Soprano Titanium Certified Clinic (Alma Lasers), Exosome Therapy (ExoCoBio), KFERA, KOAT, and ASLS. Head Director Seung-Jae Baek has performed 15,000+ procedures using KFDA-approved devices, with specialized training from Solta Medical, Merz, and Sofwave Medical.
₩159,000-1,990,000 at RE:BERRY Myeongdong for genuine KFDA-approved devices, which is 50-70% less than identical treatments in the US.
| Device | US Average | RE:BERRY Price | Savings |
|---|---|---|---|
| Thermage FLX 600 shots | $3,500-5,000 | ₩1,990,000 (~$1,440) | 60% |
| Ultherapy Prime 200 shots | $2,000-3,000 | ₩980,000 (~$710) | 65% |
| PicoSure Toning | $400-600 | ₩159,000 (~$115) | 70% |
50-70% lower pricing at RE:BERRY Myeongdong reflects Korea’s competitive density of 14,000+ aesthetic clinics nationwide, not lower device quality.
| Cost Factor | Korea | US/UK/Japan |
|---|---|---|
| Clinic Density | 14,000+ (competitive pricing) | Lower density, higher margins |
| Device Supply Chain | Direct from Korean manufacturers | Import + distribution markup |
| Consumable Cost | Local production (Potenza, Juvelook) | Export pricing + customs |
₩249,000 for genuine Potenza RF at RE:BERRY Myeongdong versus ₩80,000-120,000 at clinics using counterfeit tips that deliver 35-62% less energy.
| Factor | RE:BERRY (Genuine KFDA) | Counterfeit Device Clinic |
|---|---|---|
| Thermage Tip Cost | ₩300,000-400,000 (genuine) | ₩30,000-50,000 (fake) |
| Energy Delivery | 100% calibrated | 35-62% of specs |
| Complication Rate | Under 0.5% | 3-5x higher |
5 KFDA-approved lifting platforms at RE:BERRY Myeongdong, each targeting different tissue depths for comprehensive facial tightening.
| Device | Technology | Depth | Price |
|---|---|---|---|
| Ultherapy Prime | Focused Ultrasound | 4.5mm SMAS | ₩490,000/100 shots |
| Sofwave SUPERB | 3D HIFU | 1.5mm dermis | ₩990,000/100 shots |
| Onda | Microwave | Subcutaneous fat | ₩1,550,000/80kJ |
₩1,990,000 Thermage FLX for volumetric tightening versus ₩490,000/100 shots Ultherapy Prime for deep SMAS lifting at RE:BERRY Myeongdong — different mechanisms for different concerns.
| Feature | Thermage FLX | Ultherapy Prime |
|---|---|---|
| Technology | Radiofrequency | Focused Ultrasound |
| Target Depth | Dermis + subcutaneous | SMAS at 4.5mm |
| Duration | 12-18 months | 12-24 months |
| Downtime | None | Mild tingling 24h |
6 KFDA-approved injectable products at RE:BERRY Myeongdong, all Korean-developed and batch-verified for authenticity.
| Product | Manufacturer | KFDA Approval |
|---|---|---|
| Rejuran Healer | Pharma Research Products | Polynucleotide PN |
| Juvelook | HansBiomed | PLLA skin booster |
| Re2O | Korean biotech | Skin rejuvenation |
30+ KFDA-approved devices at RE:BERRY Myeongdong versus 8-15 at typical Seoul clinics and 15-20 at premium Gangnam clinics.
| Feature | RE:BERRY (Boutique) | Large Chain Clinic |
|---|---|---|
| KFDA-Approved Devices | 30+ | 8-15 |
| Manufacturer Certifications | 8 | 1-3 |
| Consumable Verification | Opened in front of patient | Pre-attached |
| Device Purchase Source | Authorized Korean distributors | Varies |
3-5x higher complication rates at clinics using non-KFDA-approved devices versus under 0.5% at RE:BERRY Myeongdong.
| Risk Factor | RE:BERRY (KFDA-Certified) | Non-Certified Clinic |
|---|---|---|
| Device Energy Accuracy | 100% of manufacturer specs | 35-62% of specs |
| Complication Rate | Under 0.5% | 1.5-2.5% |
| Result Consistency | Clinical trial-matched | Unpredictable |
3 verification methods at RE:BERRY Myeongdong: (1) KFDA registration stickers visible on every device, (2) 8 manufacturer certifications displayed in the clinic lobby, and (3) single-use consumables opened in front of each patient with holographic authenticity stickers. Thermage FLX tips carry Solta Medical serial numbers, Potenza RF tips are factory-sealed single-use, and Ultherapy transducers are scanned before every treatment.
Under 0.5% complication rate across 30,000+ treatments at RE:BERRY Myeongdong using genuine KFDA-approved devices. Emergency protocols include on-site medical intervention by 2 licensed physicians, direct manufacturer support channels for device-specific issues, and referral to Severance Hospital (5 minutes away). Full treatment records with device serial numbers support insurance or medical claims.
2-5 days depending on treatment complexity at RE:BERRY Myeongdong. Single treatments (laser toning, botox, skin boosters) require 1 day with zero downtime. Combination lifting packages need 2-3 days. Multi-treatment plans spanning 3-5 days maximize the device variety available at our KFDA-approved clinic.
100% of patients receive 2 scheduled follow-ups at 48 hours and 7 days post-treatment from RE:BERRY Myeongdong, plus written aftercare instructions in 8 languages. International patients who return home continue receiving remote aftercare via WhatsApp or LINE for up to 30 days. Every treatment record includes the specific KFDA-approved device model and serial number used for your medical documentation.
Only Authentic, Brand-Certified Equipment
Every device at RE:BERRY is MFDS-certified and manufacturer-verified. We never use counterfeit or grey-market equipment.
Experience Treatment at a Genuine KFDA-Approved Devices Clinic
Book your consultation at RE:BERRY Myeongdong — 30+ KFDA-approved devices, 8 manufacturer certifications, 2 licensed physicians. 2 minutes from Myeongdong Station, open 365 days, 8 languages.
Book Your Treatment at Our KFDA-Approved Devices Clinic
Fill out the form and our multilingual team will confirm within 2 hours via your preferred contact method.
- ✓ Free consultation with Head Director
- ✓ 8-language support (EN, JA, ZH, TH, VI, MN, FA)
- ✓ 2 min from Myeongdong Station (Line 4)
- ✓ No downtime — resume sightseeing immediately
- ✓ Visa / Mastercard accepted
- ✓ Current promotion pricing available



.webp)