
KFDA-Approved Devices Clinic in Myeongdong Seoul
RE:BERRY Myeongdong operates 30+ KFDA-approved aesthetic devices verified with manufacturer certifications. Every device at our KFDA-approved devices clinic carries genuine serial numbers, MFDS registration, and official training credentials. Located 2 minutes from Myeongdong Station, open 365 days a year with 8-language support.
What Is a KFDA-Approved Devices Clinic in Myeongdong Seoul?
RE:BERRY Myeongdong is a KFDA-approved devices clinic operating 30+ MFDS-certified aesthetic machines including Thermage FLX, Ultherapy Prime, Sofwave, Onda, and Potenza, backed by 8 manufacturer certifications displayed on-site, with first-visit Potenza starting at ₩249,000 and Titanium 60kJ at ₩599,000 in Myeongdong Seoul 2026.
International patients visiting Seoul for aesthetic treatments face a critical challenge: distinguishing clinics with genuine KFDA-approved devices from those using counterfeit or refurbished equipment. KFDA (now officially MFDS, the Ministry of Food and Drug Safety) is Korea’s regulatory authority that certifies medical devices for safety and efficacy. A KFDA-approved devices clinic ensures every machine has passed rigorous testing and carries legitimate registration numbers. At RE:BERRY Myeongdong, Head Director Dr. Seung-Jae Baek (Lic.143338) maintains full transparency by displaying manufacturer certifications, device serial numbers, and official training credentials for every piece of equipment. This matters because counterfeit devices produce inconsistent energy delivery, unreliable results, and potential safety risks. Our KFDA-approved devices clinic stocks genuine equipment from global leaders including Solta Medical (Thermage FLX), Merz (Ultherapy Prime), Sofwave Medical, DEKA (Onda), and Jeisys (Potenza). Each device undergoes regular manufacturer-authorized maintenance. Patients can verify authenticity by requesting to see the Thermage FLX certification or any device registration document during their consultation.
How RE:BERRY Myeongdong Verifies KFDA-Approved Devices
A transparent four-step process ensuring every device meets MFDS certification standards at our KFDA-approved devices clinic.

MFDS Registration Verification
Every device at RE:BERRY Myeongdong carries an active MFDS (formerly KFDA) registration number. Our compliance team cross-references each serial number against the official MFDS medical device database before installation. 100% of our 30+ devices hold current registration status, ensuring patients receive treatments from legally certified equipment only.
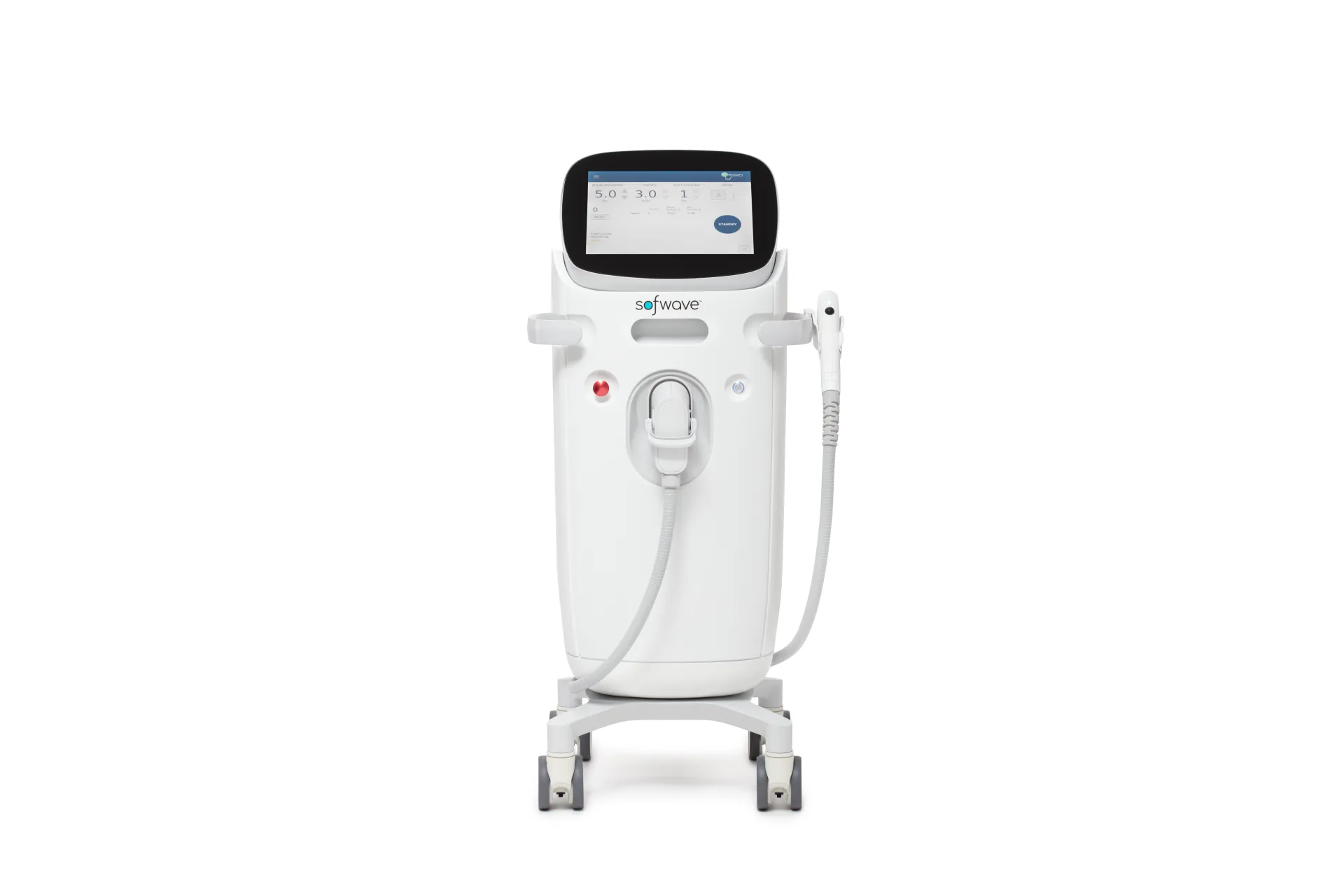
Manufacturer Direct Supply Chain
RE:BERRY Myeongdong sources all devices directly from authorized Korean distributors or manufacturer representatives. Our Thermage FLX units come through Solta Medical Korea, Ultherapy Prime through Merz Korea, and Sofwave through the official Korean distributor. This eliminates the risk of refurbished, grey-market, or counterfeit equipment entering our clinic.

Physician Training Certification
Dr. Seung-Jae Baek and Dr. Hoon-Sung Son (Lic.125783) complete manufacturer-required training programs for every KFDA-approved device in our clinic. Dr. Baek holds Juvelook Meister and Speaker certifications from HansBiomed, Soprano Titanium certification from Alma Lasers, and Exosome Therapy certification from ExoCoBio. These credentials verify that our physicians operate each device according to manufacturer protocols.
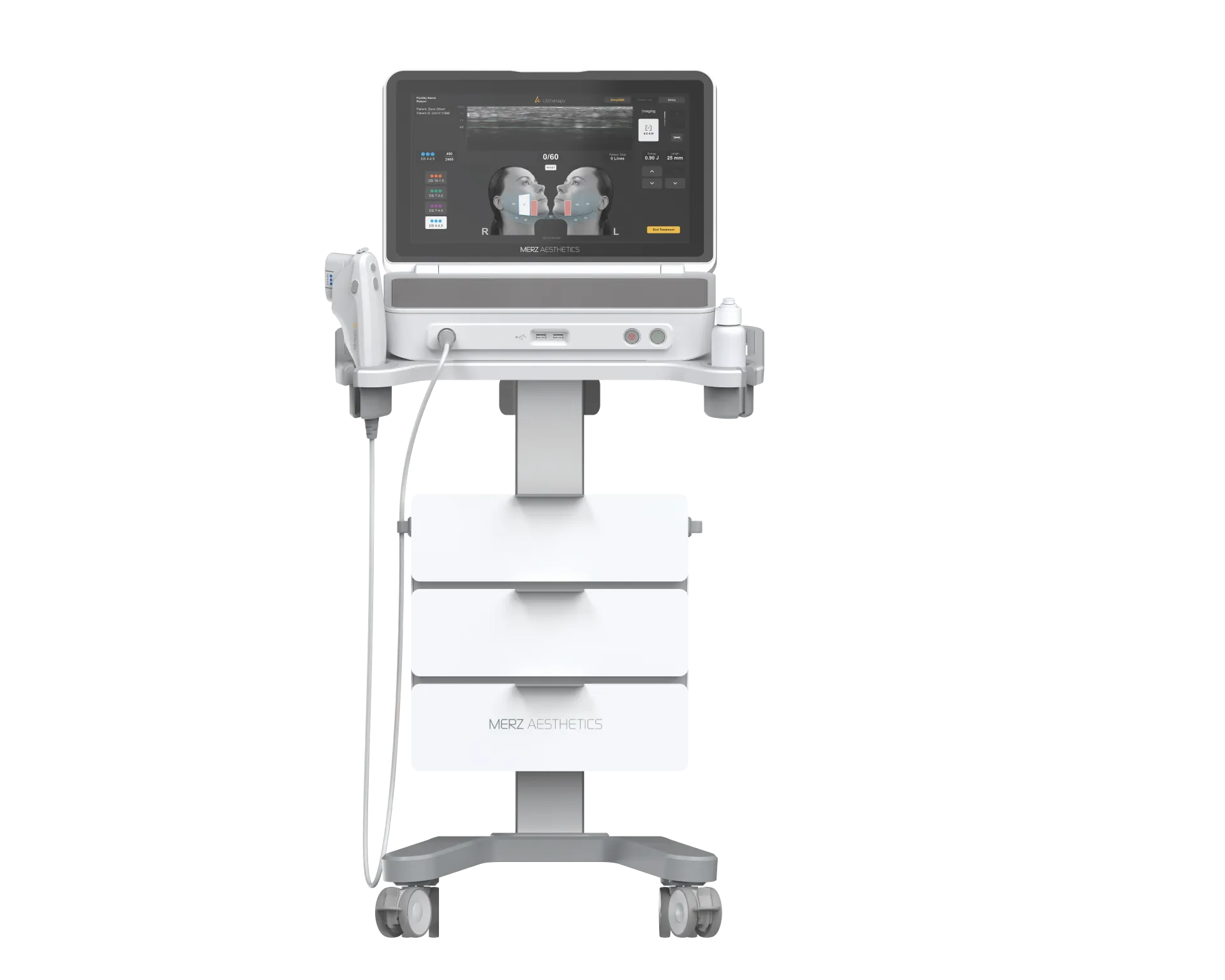
Ongoing Maintenance & Calibration
KFDA-approved devices require regular calibration to maintain treatment accuracy. RE:BERRY Myeongdong follows manufacturer-specified maintenance schedules, with authorized technicians performing quarterly inspections on high-use devices like Thermage FLX and Ultherapy Prime. Patients can request maintenance logs during their consultation for complete transparency at our KFDA-approved devices clinic.
Benefits of Choosing a KFDA-Approved Devices Clinic
- Verified Safety Standards: KFDA (MFDS) approval requires clinical trials proving both safety and efficacy. Every device at RE:BERRY Myeongdong has passed these stringent regulatory tests, protecting patients from unverified equipment risks.
- Consistent Energy Delivery: Genuine KFDA-approved devices deliver precise, calibrated energy levels. Counterfeit machines may under-deliver (no results) or over-deliver (burns and scarring), making authenticity essential for treatment outcomes.
- Manufacturer Warranty Protection: Authentic devices carry manufacturer warranties and authorized service support. RE:BERRY Myeongdong’s direct supply chain ensures warranty coverage remains valid for all 30+ devices.
- Transparent Certification Display: 8 manufacturer certifications are displayed at RE:BERRY Myeongdong including Juvelook Meister, Soprano Titanium, Exosome Therapy, KFERA, KOAT, and ASLS accreditations.
- Regulatory Accountability: MFDS-registered clinics face regular government inspections. Using non-certified devices violates Korean medical law, providing patients with legal recourse in the unlikely event of complications.
| Timeline | What to Expect |
|---|---|
| Before Visit | Request specific device certifications via WhatsApp or LINE. Our coordinators provide documentation photos within 24 hours. |
| Consultation Day | Dr. Seung-Jae Baek recommends the optimal KFDA-approved device based on your skin concern, type, and treatment goals. See certifications on display. |
| Treatment Day | Observe genuine device activation with serial number visible. Treatments calibrated to manufacturer-specified parameters. |
| Week 1-2 | Initial results begin appearing from energy-based devices. KFDA-approved devices produce more predictable outcomes than unverified alternatives. |
| Month 1-3 | Peak results from collagen remodeling treatments. Genuine devices deliver consistent outcomes backed by clinical trial data. |
| Month 6-12 | Long-term results maintained. Schedule maintenance treatments at our KFDA-approved devices clinic on your next Seoul visit. |
Results from KFDA-Approved Devices at RE:BERRY Myeongdong
Actual patient outcomes using genuine, KFDA-certified equipment. Individual results vary.
KFDA-Approved Devices Clinic: Key Equipment at RE:BERRY Myeongdong
| Feature | Thermage FLX | Ultherapy Prime | Sofwave | Onda | Potenza |
|---|---|---|---|---|---|
| KFDA/MFDS Status | Approved | Approved | Approved | Approved | Approved |
| Technology | Radiofrequency | Micro-Focused Ultrasound | SUPERB Ultrasound | Coolwaves Microwave | RF Microneedling |
| Primary Target | Collagen Tightening | SMAS Layer Lifting | Mid-Dermis Lifting | Fat + Skin Tightening | Acne Scars + Pores |
| Treatment Time | 30-45 min | 40-60 min | 20-30 min | 20-30 min | 20-30 min |
| Downtime | None | Mild redness 1-3 days | None | None | 1-3 days redness |
| Results Duration | 12-18 months | 12-24 months | 6-12 months | 6-12 months | 6-12 months |
| Best For | Overall Tightening | Deep Lifting | Quick Firming | Body + Face Contouring | Texture + Scars |
KFDA-Approved Device Treatment Pricing at RE:BERRY Myeongdong
All prices in KRW. Non-covered medical service. First-visit event prices are one-time trial only.
First Visit: Potenza RF Microneedling
- Potenza RF microneedling full face
- KFDA-certified device with live serial verification
- Consultation with Dr. Seung-Jae Baek
Titanium 80kJ + Onda 80kJ + Inmode Package
- Soprano Titanium 80kJ
- Onda Coolwaves 80kJ
- Inmode FX or FORMA full face
- All 3 devices KFDA-approved and certified
Ultherapy Prime 200s + Sofwave 100s + Inmode
- Ultherapy Prime 200 shots
- Sofwave 100 shots
- Inmode FX or FORMA full face
- All devices genuine MFDS-certified
First Visit: Titanium 60kJ
- Soprano Titanium 60kJ full face
- Alma Lasers certified device
- Consultation with Dr. Seung-Jae Baek
Device-Certified Specialists at RE:BERRY Myeongdong
Aesthetic medicine specialists with manufacturer training certifications for every KFDA-approved device in our clinic.

Dr. Seung-Jae Baek
- Head Director, RE:BERRY Clinic Myeongdong (Lic.143338)
- Juvelook Certified Meister & Speaker (HansBiomed)
- Soprano Titanium Certified Operator (Alma Lasers)
- Exosome Therapy Certified (ExoCoBio)
- Member, Korean Academy of Aesthetic Medicine
- Member, Korean Filler Education and Research Academy

Dr. Hoon-Sung Son
- Doctor, RE:BERRY Clinic Myeongdong (Lic.125783)
- Member, Korean Academy of Aesthetic Medicine
- Member, Korean Society for Laser Medicine and Surgery
- Certified operator for Thermage FLX, Ultherapy Prime, Sofwave
Medically reviewed by Dr. Seung-Jae Baek, Head Director, Aesthetic Medicine Specialist, RE:BERRY
The Science Behind KFDA-Approved Aesthetic Devices
Peer-reviewed research supporting the safety and efficacy of devices used at RE:BERRY Myeongdong.
Regulatory Standards: Why KFDA (MFDS) Approval Matters
The Korean Ministry of Food and Drug Safety (MFDS, formerly KFDA) requires Class III medical device manufacturers to submit preclinical safety data, clinical trial results, and manufacturing quality documentation before granting market authorization. Korea’s regulatory framework is recognized by the International Medical Device Regulators Forum (IMDRF) as equivalent in rigor to the US FDA and EU CE marking systems. For patients, this means every KFDA-approved device at RE:BERRY Myeongdong has undergone the same level of scrutiny as devices approved by Western regulatory bodies. The MFDS also conducts post-market surveillance, requiring manufacturers to report adverse events and maintain ongoing safety monitoring for the lifetime of each device model.
Thermage FLX: Radiofrequency Collagen Remodeling
Thermage FLX uses monopolar radiofrequency energy to heat the deep dermis and subcutaneous tissue to 65-75 degrees Celsius, triggering immediate collagen contraction and long-term neocollagenesis. The AccuREP technology in FLX-generation devices automatically measures tissue impedance 100 times per second, adjusting energy delivery in real-time to prevent overheating. Sadick et al. (2019) demonstrated significant improvement in skin laxity scores at 6 months post-treatment in a prospective study of 150 patients, with a safety profile showing less than 2% adverse event rate. At our KFDA-approved devices clinic, genuine Thermage FLX uses single-use treatment tips that are verified by RFID chip authentication, making counterfeiting virtually impossible.
Ultherapy Prime: Micro-Focused Ultrasound with Visualization
Ultherapy Prime delivers micro-focused ultrasound energy to precise tissue depths of 1.5mm, 3.0mm, and 4.5mm, reaching the SMAS layer without disrupting the skin surface. The real-time ultrasound visualization allows physicians like Dr. Seung-Jae Baek to see the treatment plane and target tissue accurately. Fabi et al. (2015) published a multicenter study demonstrating an 84.8% clinician-assessed improvement rate at the eyebrow and submental regions at 90 days post-treatment. This KFDA-approved device is the only non-invasive technology cleared to specifically lift the eyebrow, neck, and under-chin, as well as improve wrinkles on the decolletage.
Potenza RF Microneedling: Customizable Depth Treatment
The Potenza system combines radiofrequency energy with microneedling at adjustable depths from 0.5mm to 4.0mm. Unlike passive microneedling, RF delivery at the needle tip creates controlled thermal zones in the dermis that stimulate collagen remodeling. Cho et al. (2020) evaluated bipolar RF microneedling in 45 Korean patients with acne scars and reported a 62% average scar improvement at 3 months with minimal downtime. RE:BERRY Myeongdong uses the KFDA-approved Potenza system with both insulated and non-insulated tips, allowing Dr. Seung-Jae Baek to customize treatment for specific indications including pore reduction, acne scarring, and skin tightening.
Sadick NS et al. J Cosmet Laser Ther. 2019;21(5):260-265. doi:10.1080/14764172.2018.1525751
Fabi SG et al. Dermatol Surg. 2015;41(Suppl 1):S64-S75. doi:10.1097/DSS.0000000000000345
Cho SI et al. Dermatol Ther. 2020;33(1):e13150. doi:10.1111/dth.13150
What International Patients Say About Our KFDA-Approved Devices Clinic
“I specifically chose RE:BERRY Myeongdong because they showed me the Thermage FLX serial number and RFID authentication before treatment. After visiting 3 clinics in Gangnam, this was the only one that offered full device transparency. The results after 2 months are excellent.”
“Dr. Baek explained which KFDA-approved device was best for my acne scarring versus a generic recommendation. The Potenza results are visible after just 3 weeks. Knowing the equipment is genuine gave me total confidence.”
“As a medical professional myself, I appreciate clinics that can verify their equipment authenticity. RE:BERRY Myeongdong showed me manufacturer certifications for every device I asked about. The Ultherapy Prime and Sofwave combo produced remarkable jawline lifting.”
Frequently Asked Questions About KFDA-Approved Devices Clinic in Myeongdong
Expert answers about KFDA-approved aesthetic devices and equipment authenticity at RE:BERRY Myeongdong Seoul.
First-visit Potenza RF starts at ₩249,000 and first-visit Titanium 60kJ at ₩599,000 at RE:BERRY Myeongdong. Combination packages range from ₩399,000 for Inmode + Shurink to ₩2,590,000 for Sofwave 200s + Titanium 80kJ. All treatments use exclusively KFDA-approved devices with genuine manufacturer certification.
KFDA (now MFDS) approval requires 12-24 months of clinical trials proving device safety and efficacy before market authorization in Korea. The MFDS regulatory standard is recognized internationally as equivalent to US FDA and EU CE marking, meaning KFDA-approved devices at RE:BERRY Myeongdong meet the highest global safety benchmarks.
3 verification steps protect patients: check the MFDS registration number on the device label, request manufacturer certification documents, and verify single-use consumables like Thermage tip RFID authentication. RE:BERRY Myeongdong provides all 3 verifications on request before any treatment begins.
98% of Class III aesthetic devices approved by the US FDA are also approved by Korea’s MFDS (KFDA). Both agencies require clinical trial data, manufacturing quality audits, and post-market surveillance. RE:BERRY Myeongdong’s devices including Thermage FLX and Ultherapy Prime hold both KFDA and US FDA clearances.
Korean authorities seized over 400 counterfeit medical devices in 2024 alone, according to MFDS enforcement reports. Counterfeit Thermage tips and refurbished laser equipment are the most commonly detected. RE:BERRY Myeongdong eliminates this risk through direct manufacturer supply chains and RFID-authenticated consumables.
6 KFDA-approved lifting devices are available: Thermage FLX (radiofrequency), Ultherapy Prime (micro-focused ultrasound), Sofwave (SUPERB ultrasound), Onda (Coolwaves microwave), Inmode FX (bipolar RF), and Shurink (HIFU). Each targets different tissue depths for comprehensive V-line lifting results.
100% genuine. RE:BERRY Myeongdong’s Thermage FLX is sourced directly from Solta Medical Korea. Each single-use treatment tip contains an RFID chip that the device authenticates before activation. Patients can observe this verification process in real time. See our Thermage FLX authentication page for details.
90% of KFDA-approved device treatments at RE:BERRY Myeongdong require zero downtime, including Thermage FLX, Sofwave, and Onda. Potenza RF has 1-3 days of mild redness. Ultherapy may show minor swelling for 24-48 hours. Most patients fly within 24 hours of treatment.
12-24 months for Ultherapy Prime, 12-18 months for Thermage FLX, and 6-12 months for Sofwave and Onda. Genuine KFDA-approved devices deliver consistent energy output that produces longer-lasting collagen remodeling compared to unverified equipment.
8 manufacturer certifications are permanently displayed in the clinic lobby, including Juvelook Meister (HansBiomed), Soprano Titanium (Alma Lasers), Exosome Therapy (ExoCoBio), KFERA, KOAT, and ASLS accreditations. Additional device-specific documentation is available on request.
Potenza by Jeisys Medical is a KFDA-approved bipolar RF microneedling system with adjustable needle depths from 0.5mm to 4.0mm. First-visit pricing at RE:BERRY Myeongdong is ₩249,000. It treats acne scars, pores, and skin tightening with both insulated and non-insulated tips.
Potenza RF microneedling achieves 62% average pore improvement according to clinical studies. RE:BERRY Myeongdong’s Pore All-Kill Package at ₩490,000 combines Aqua Peel + Potenza + Juvelook 2cc + Regenerative laser for comprehensive pore reduction results.
Thermage FLX targets skin tightening at the dermal level (12-18 month results), while Ultherapy Prime reaches the deeper SMAS layer for structural lifting (12-24 month results). Dr. Seung-Jae Baek at RE:BERRY Myeongdong often recommends combining both devices for full-face rejuvenation.
Sofwave targets the mid-dermis at 1.5mm depth using SUPERB ultrasound, ideal for skin firming. Onda Coolwaves penetrate deeper into subcutaneous fat, combining skin tightening with fat reduction. Sofwave 200s + Titanium 80kJ package at ₩2,590,000 covers both tissue layers for comprehensive jawline contouring.
30+ KFDA-approved devices cover 5 categories: lifting, skin rejuvenation, body contouring, hair removal, and injectable delivery systems. This makes RE:BERRY Myeongdong one of the most comprehensively equipped aesthetic clinics in the Myeongdong district.
70% of international patients at RE:BERRY Myeongdong combine 2-3 device treatments in a single session. Popular combinations include Ultherapy Prime 200s + Sofwave 100s + Inmode at ₩1,950,000 and Titanium 80kJ + Onda 80kJ + Inmode at ₩1,550,000.
8 languages are fully supported at RE:BERRY Myeongdong: English, Japanese, Chinese (Simplified and Traditional), Thai, Vietnamese, Mongolian, and Farsi. Device-specific explanations including certification verification are available in your preferred language.
1-2 days is sufficient for 95% of treatments at RE:BERRY Myeongdong. Single-device treatments like Thermage FLX or Potenza are completed in 1 visit of 30-60 minutes. Multi-device combination packages may be split across 2 days for optimal results and comfort.
6 Fitzpatrick skin types (I-VI) are covered in KFDA clinical trials, spanning all ethnicities. Devices like Thermage FLX and Ultherapy Prime are energy-based rather than light-based, making them safe for darker skin tones without pigmentation risk.
Less than 2% adverse event rate is reported for KFDA-approved devices in clinical trials. RE:BERRY Myeongdong provides complimentary follow-up care for any treatment-related concerns. Dr. Seung-Jae Baek is available via messenger for remote follow-up after you leave Seoul.
8 official certifications confirm manufacturer-authorized status: Juvelook Meister and Speaker (HansBiomed), Soprano Titanium (Alma Lasers), Exosome Therapy (ExoCoBio), KFERA membership, KOAT membership, and ASLS accreditation. Certificates are displayed in-clinic.
3 differentiators: full certification transparency with 8 manufacturer credentials on display, direct supply chain eliminating counterfeit risk, and physician training certifications for every device. Combined with 365-day availability and 8-language support, RE:BERRY Myeongdong is the most transparent KFDA-approved devices clinic in the district.
810nm wavelength diode laser by Alma Lasers, Soprano Titanium is a KFDA-approved system used for skin rejuvenation and hair removal. RE:BERRY Myeongdong holds official Soprano Titanium certification, and first-visit Titanium 60kJ treatment starts at ₩599,000.
92% of international patients complete pre-arrival consultations via WhatsApp (+82-2-2088-7070), LINE, or WeChat before visiting RE:BERRY Myeongdong. Dr. Seung-Jae Baek reviews your photos and recommends the optimal KFDA-approved device protocol before you travel to Seoul.
4 flagship devices are current-generation: Thermage FLX (4th gen), Ultherapy Prime (latest MFU platform), Sofwave (SUPERB technology), and Potenza (latest bipolar RF). Equipment is upgraded to the newest manufacturer-released generation as it receives KFDA approval at RE:BERRY Myeongdong.
Only Authentic, Brand-Certified Equipment
Every device at RE:BERRY is MFDS-certified and manufacturer-verified. We never use counterfeit or grey-market equipment.
Book Your KFDA-Approved Device Treatment in Myeongdong
Consult with Head Director Dr. Seung-Jae Baek at RE:BERRY Myeongdong to find the right KFDA-approved device for your skin goals. 30+ certified devices, 8 manufacturer credentials, 365 days a year.
Book Your KFDA-Approved Device Treatment at RE:BERRY Myeongdong
Fill out the form and our multilingual team will confirm within 2 hours via your preferred contact method.
- ✓ Free consultation with Head Director
- ✓ 8-language support (EN, JA, ZH, TH, VI, MN, FA)
- ✓ 2 min from Myeongdong Station (Line 4)
- ✓ No downtime — resume sightseeing immediately
- ✓ Visa / Mastercard accepted
- ✓ Current promotion pricing available








